PuTTY plays a significant role in secure transfer and connectivity from the local host to a remote system. The native file format is .ppk files. The private key must be in the native form of the application if you are using PuTTY.
The Cloud computing platform is a core part of EC2 and generates the PEM file format; before connecting to Linux, the user converts the file into a .ppk file format.
What is .pem File Format?
In 1993 IETC defines “privacy-enhanced mail” known as .PEM file format. It contains multiple instances. Suffixes like “.pem”, a “.cer” or a “.crt” or a”.key” are used to store Pem data in files. Cryptographic keys, certificates, and other data are stored and sent by the extension .PEM file format.
The PEM format defines three elements:
- A one-line header, consisting of “—–BEGIN”, a label, and “—–“.
- Base64 encoded binary data.
- A one-line footer, consisting of “—–END”, a label, and “—–“.
.PEM file format is safe for ASCII and rich-text documents, such as emails.
Example of a certificate stored:

Multiple Messages
The PEM file may contain multiple messages; it can combine the certificate or provide the certificate chain and the private key in a single file.
What is .PPK file format?
The private keys generated by the program are stored in .PPK files. PuTTY develops the Private Key file, and the key file authentication is stored in .PPK files make it serves as a computer maker.
The steps for authentication .PPK files are as follows:
- Choose the connection.
- SSH → Auth (Putty Configuration Menu).
- Click → Browse → Enable Option “Private Key File”.
Note: .PPK files can be opened for Windows, Linux, and MAC OS.
How to convert PEM to PPK file? (Windows)
The steps are as follows:
1. Start menu → All Programs → PuTTY → PuTTYkey
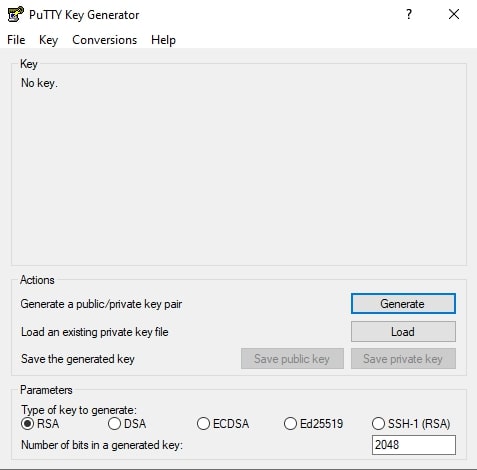
2. Select option → RSA
RSA is used to transmit data securely and is known as a public-key cryptosystem. Select the option SSH-2 RSA for the old version of PuTTY.

3. Click → Load
It only shows the .ppk files extension, as Putty supports the native file format.
The user must follow the sub-step:
- Dropdown bar → All files
Display All key files included in the .pem file.
4. Select → .pem file
As mentioned earlier, the user must select the specific file that Putty is used for SSH connectivity.
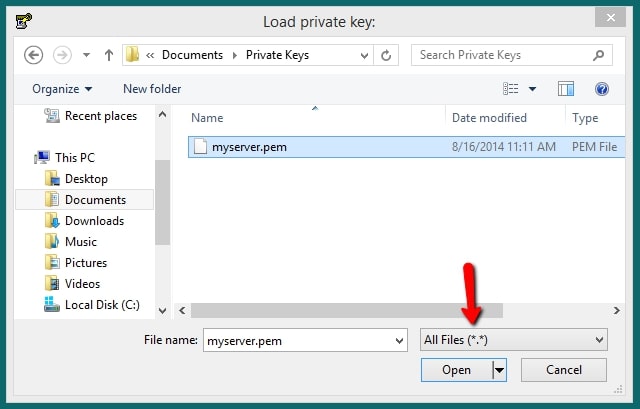
- Click → Open
- To confirm, click → Ok
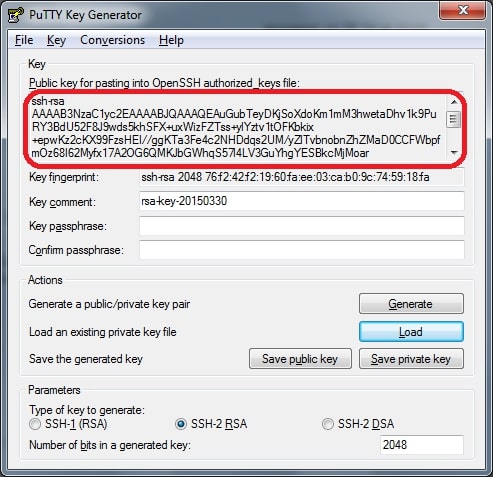
5. Click → ‘Save private key.’
6. Putty prompt warning for saving the key without a Passphrase. Hit “Yes” on it.
7. Putty will automatically add the .ppk file extension when the name is given to a file.
Note: Users can connect their local machine with remote servers when the file is converted to a PuTTY-compatible format.
How to convert PPK file to PEM in macOS?
First, you must install Homebrew to convert .ppk to .pem file in macOS. After that, you need to follow below steps:
- First, install the PuTTY.
brew install putty - Second, Convert Putty Key to Open SSH.
puttygen puttykey.ppk -O private-openssh -o privatekey.pem
How to convert PPK file to PEM file? (Linux)
First, install the Putty application on the Unix machines. Then run the PuTTYgen command and type the below command.
$ sudo puttygen pemKey.pem -o ppkKey.ppk -O private
